Recombinant antibodies are highly specific detection probes in research, diagnostics, and have emerged over the last two decades as the fastest growing class of therapeutic proteins. Antibody generation has been dramatically accelerated by in vitro selection systems, particularly phage display.
Various enhancements to recombinant production systems have been developed, ranging from gram-negative and positive bacteria, filamentous yeasts and fungi, insect cell lines, mammalian cells to transgenic plants and animals. You can also browse https://www.bosterbio.com/featured-products to know more about antibodies.
Currently, almost all therapeutic antibodies are still made in mammalian cell lines to reduce the risk of immunogenicity due to changes in non-human glycosylation patterns.
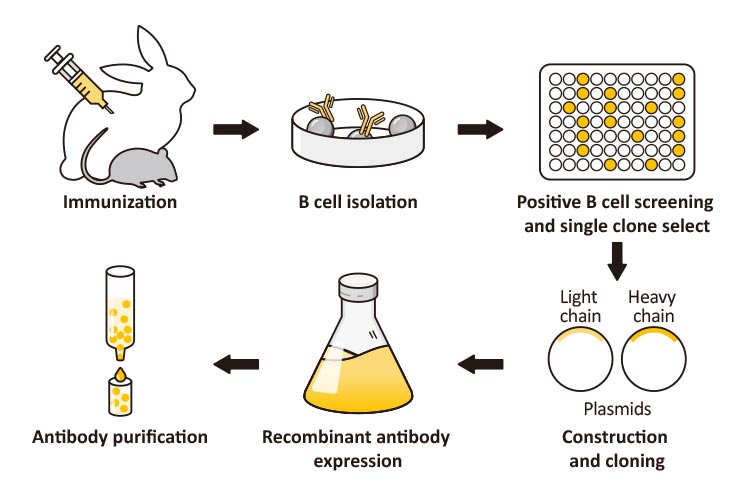
Image Source: Google
However, recent developments in glycosylated injected yeast, insect cell lines and transgenic plants promise to produce antibodies with "human-like" post-translational modifications.
In addition, smaller antibody fragments, including bispecific antibodies without any glycosylation, have been successfully produced in bacteria and clinically tested. The first therapeutic antibody products from non-mammal sources are expected in the years to come. In this review, we focus on modern antibody production systems, including their application to various applications.
Antibodies are used today for a variety of applications in research, diagnosis, and therapy. They are used in many standard tests such as immunoblot, flow cytometry, or immunohistochemistry.
In addition, there is a great need in the emerging field of proteome research for binding to various protein antigens and splice variants. In addition, recombinant antibodies are used to diagnose various pathogens or toxins.